Plasma Files: liquid gold, business and incidents
It allows saving lives. Severe bleeding and accidents, major burns, rare diseases: plasma makes it possible to fight these formidable threats and produce life-saving drugs like albumin or immunoglobulins. Even during the current epidemic, we find ourselves hoping to find a therapy that can fight Covid thanks to plasma. But precisely because donating plasma is an act of altruism, donating it should be completely safe. Is it? Il Fatto Quotidiano has conducted an eight-month journalistic investigation in partnership with seven international media outlets, coordinated by the non-profit organization The Signals Network which provides support to selected whistleblowers.
We had exclusive access to hundreds of internal documents emitted by Haemonetics, an American multinational company which produces and markets one of the most widely used plasma donation technologies: the machines and disposable kits for apheresis. There are three giants competing for this market worldwide: the American Haemonetics, the Japanese Terumo and the German Fresenius.
From Bolzano to Palermo, Haemonetics machines are widely used in Italy to collect plasma on a regular basis and, in recent weeks, they have also been used by major Italian hospitals – such as Pisa, San Camillo in Rome, Cotugno in Naples, Policlinico in Bari – to collect plasma from patients who have recovered from Covid, the so-called convalescent plasma.
The internal documents of the American multinational – shared with us by some whistleblowers – allow for the first time piecing together certain incidents that have occurred with these machines throughout the world, Italy included: some of those incidents could potentially constitute a risk for donors.
Liquid gold
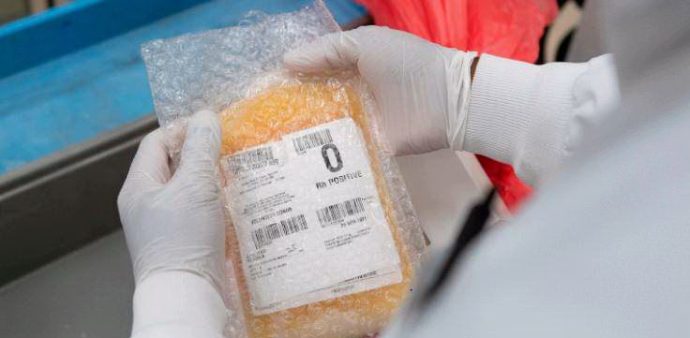
They call it “liquid gold” for how precious it is, and for its vital importance in medicine. Plasma is what allows blood to flow, a yellowish liquid in which blood cells such as red and white blood cells and platelets are suspended. If you take those cells out of the plasma, you are left with a yellowish liquid that is mostly water – about 92%. The rest is made up of proteins, minerals, enzymes and immunoglobulins.
The word ‘apheresis’ comes from Greek, and literally means ‘taking away’. It can be therapeutic apheresis – with the purpose of curing diseases caused by abnormal blood cells – or productive apheresis, which allows selectively donating plasma, plasma and platelets, red blood cells and platelets, etc. While for a blood donation all you need is a syringe, productive apheresis requires an apheresis machine which processes large volumes of blood, separates the different blood components, and returns the remainder to the donor, usually through the same needle whereby the blood was withdrawn.
The machine consists of a motor, pumps, sensors, the software that controls the process, and finally a disposable kit in which the donor’s blood circulates. The kit consists of a bowl with a centrifuge that separates the different components of the blood, tubes and collection bags.
Depending on the blood components to be extracted, different disposable kits are used. Among the companies that produce and market apheresis machines, the US company Haemonetics – with its MCS+ and PCS2 plasma collection machines – is one of the most important in the world.
France: The Whistleblowers
The Haemonetics case exploded in France, in 2017. For years, three whistleblowers, Alexandre Berthelot, Philippe Urrecho and Guylain Cabantous, had been alerting the French health authorities on the risks of these machines and their disposable kits.
According to them, the problem stems from “black particles” which, in some cases, are produced during the apheresis process and end up in the collected plasma. They are particles whose nature and long-term effects are not clear, especially for those donors who donate regularly. What do these particles consist of? According to the company, they are dried blood or dried proteins, while the three whistleblowers believe this black particulate could contain potentially harmful substances emanating from friction and the overheating of the components in the centrifuge bowl.
In 2017, the French National Agency for Medicines and Health Products Safety (ANSM) established a Temporary Specialist Scientific Committee (CSST) to investigate the case.
While acknowledging that the production of large particles “is a rare phenomenon (1.8 cases out of 100,000 apheresis procedures in 2016)” and studies on the long-term effects are not available, the committee suggested: “If a reliable risk assessment cannot be obtained quickly, consideration should be given to the removal of medical devices containing movable seals (graphite/ceramic type) which may release substances known to be potentially toxic at low doses (formaldehyde, chromium VI, polycyclic aromatic hydrocarbons).” The Committee also wrote that as far as the problem with particles was concerned: “The machines marketed by Haemonetics appear to be those for which the risk seems higher“.
The turning point came in May 2018, when the three whistleblowers filed a criminal complaint against Haemonetics and the French health authorities at the Tribunal de Grande Instance in Paris. A few months later, the French health authorities suspended the use of the Haemonetics MCS+ and PCS2 machines when used with the 782HS-P-SL disposable kit. To this day, the kit is still suspended and 300 PCS2 machines have been withdrawn.
The criminal complaint filed by Berthelot, Urrecho and Guylain prompted eight plasma donors to step forward and file a criminal complaint against the multinational company. The French lawyer Alma Basic, who represents them, tells Il Fatto: “Some of the donors who filed a criminal complaint feel betrayed: for years they donated plasma as an act of pure altruism, trusting that the French institutions would protect their health, then the case of the whistleblowers led them to reconsider everything and they stopped donating“.
Italy and the World
Il Fatto Quotidiano and our international media partners contacted Haemonetics for questions and comment. The company replied: “In the past 15 years, our products have been used safely in more than 360 million plasma collection procedures globally”. The multinational company claims that the problem of “black particles” is extremely rare: particle complaints would represent “only 0.0006% of estimated total plasma collection procedures” and would not have “been associated with donors, patients or user harm”.
The internal documents in our possession and the journalistic investigations carried out on them first and foremost by Die Zeit tell of over 600 particle incidents in the last 15 years, 56 of them in Germany, and 150 in the USA, according to our US media partner, Miami Herald, in the USA.
What about Italy? In our country, Haemonetics MCS+ and PCS2 machines are widespread. They are used by the blood transfusion service in Bolzano (6 units), by the Città della Salute in Turin, by the blood transfusion centres in Modena, Parma, Piacenza, Reggio Emilia and the Azienda USL Emilia Romagna. In Tuscany, there are 144 of them (82 PCS2 and 62 MCS+). Lazio has 40 machines, Umbria 10, Campania 41 and Calabria 10.
Two watchdogs are supposed to monitor the safety of these machines: the Centro Nazionale Sangue and the Direzione generale dei dispositivi medici of the Italian Ministry of Health.
Il Fatto contacted the Italian Ministry of Health to ask some questions about Haemonetics: the Ministry never replied. Instead the Director of the Centro Nazionale Sangue, Giancarlo Maria Liumbruno, immediately replied to us that after the 782-HS-P-SL kit was suspended in France, he checked whether it had ever been marketed in Italy and learned that it had not. The US company Haemonetics confirmed to us that that kit has never been commercialised in Italy.
Thanks to our investigation, we have been able to independently verify that, between 2018 and 2019, there were at least five incidents involving the filters of the disposable kit 999F-E for plasma and platelets donation. Three of these incidents occurred at the Meyer hospital in Florence (February-March 2019), and two at the Parma hospital (January-February 2019). Both facilities told us that no harm was done to donors, but neither Meyer nor Parma provided any details on what happened.
Incidents also occurred at the Cervello Hospital in Palermo, in 2018. The head of the apheresis service, Noemi Agosto, replied: “We had problems with a haemonetics (sic) 946 disposable kit due to a faulty production that stopped us almost a year”. To our questions as to whether there had been any problems with donors, the hospital did not answer. Haemonetics also did not answer our questions on what happened in the case of incidents in Parma, at the Meyer and Cervello hospitals.
None of these incidents have been reported on the Ministry of Health’s database of medical device reports. According to our media partners, there are many incidents that have never reported in France and the US either. Are the Italian incidents we were able to verify the only ones that have occurred, or have there also been others? Are there risks for donors? Machines the use of which has been suspended in France continue to be used in Italy: the same technology appears to pose safety concerns beyond the Alps, but not on this side of the Alps. How is this possible?
This article is the result of months of investigation on hundreds of new documents provided by whistleblowers to the media partners of The Signals Network, including Bastamag (France), D
The Signals Network is a European-American non-profit organization founded by Gilles Raymond and led by Delphine Halgand-Mishra. It partners with a dozen media outlets representing a cumulative audience of 165 million readers in 7 languages. The Signals Network also provides support to selected whistleblowers.
If you are a long-term plasma donor and would like to learn more about the criminal complaint made by donors in France and/or discuss with lawyers about this, you can send an email to plasmadonors@protonmail.com.

